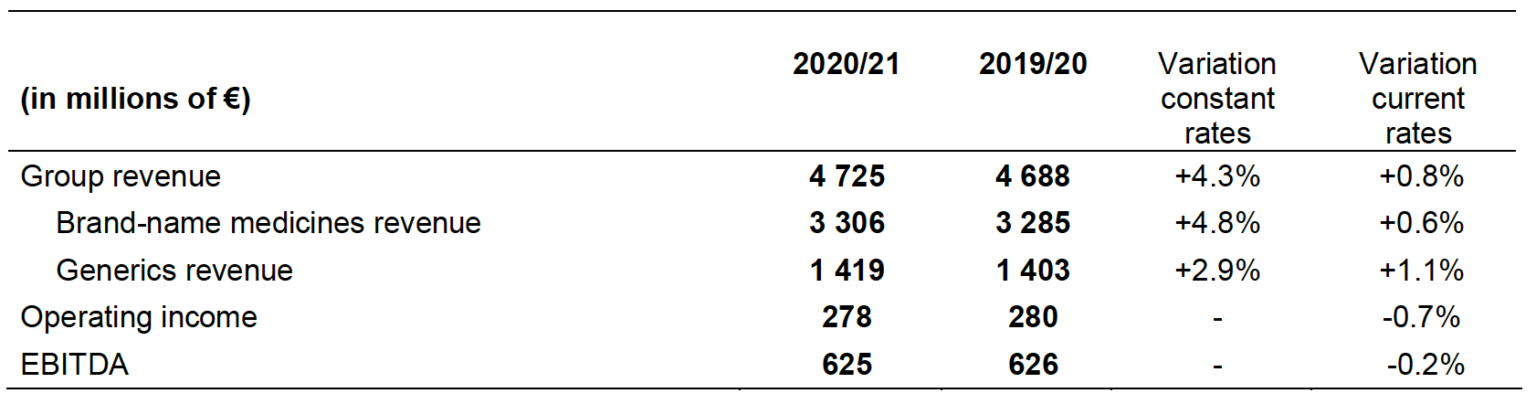
- Consolidated revenue of €4.725 billion, up +4.3% at constant rates (+0.8% at current rates)
- Revenue for brand-name medicines of €3.306 billion +4.8% at constant rates (+0.6% at current rates) and €1.419 billion for the generics +2.9% at constant rates (+1.1% at current rates)
- An accelerated oncology strategy with the acquisition of the Agios Pharmaceuticals’ oncology business in April 2021
- Sustained activity in cardiometabolism and a strong increase in sales of Daflon®
- A strengthened and balanced R&D portfolio offering hope for patients
Paris (France), January 18, 2022 – Servier, an independent global pharmaceutical group, published its results for the 2020/21 financial year, ending on September 30, 2021, and presented its research and development pipeline.
Olivier Laureau, President of Servier: “In 2020/21, the Servier Group continued to grow despite a difficult environment. We have significantly strengthened our position in oncology with the acquisition of the Agios Pharmaceuticals’ oncology business. The oncology strategy that we initiated in 2017 is now bringing results with new medicines and future indications for patients with hard-to-treat cancers. Our position in cardiometabolism remains very significant and our growth in venous diseases has been important. The Group has also initiated a deep digital transformation to accelerate its development and performance. We also strengthened our presence in key markets, particularly in Japan and the United States, while consolidating our production, bioproduction and R&D activities in France. Servier thus confirms its choice to maintain strong research and production capabilities in France, even though 96% of the Group brand-name medicines are distributed outside France. This choice contributes to the French and European health independence.”
Growth driven by an increase in sales volume
The Servier Group revenue for the 2020/21 financial year increased by 4.3% compared to 2019/20 at constant exchange rates (+0.8% at current rates) to reach €4.725 billion.
Revenue evolution is the result of a volume growth of 5.3%; i.e., €246 million. Oncology contributed €153 million to this growth, €69 million of which was generated by the acquisition of the Agios Pharmaceuticals’ oncology business. The growth in volume of the other segments was negatively impacted, due to new government measures in China.
Top 5
brand-name medicines
in 2020/21 sales
– Daflon® (€473 million)
– Diamicron® (€441 million)
– Coversyl® (€269 million)
– Preterax® (€266 million)
– Oncaspar® (€262 million)
Exchange rate variations had a negative impact of -€156 million on the 2020/21 revenue. The pressure on prices negatively impacted the revenue by -€54 million.
The revenue of brand-name medicines amounts to €3.306 billion for the 2020/21 financial year, i.e., a growth of 4.8% at constant rates (+0.6% at current rates) compared to 2019/20.
The revenue of generic medicines is up by 2.9% at constant rates (+1.1% at current rates), compared to the previous year, reaching €1.419 billion. Today, the Group has over 1,500 generic drugs covering most pathologies which are distributed worldwide by four subsidiaries: Biogaran, the leader in the generics market in France1 for the third consecutive year, EGIS in Eastern Europe, Pharlab in Brazil and Swipha in Nigeria.
The 2020/21 EBITDA remains stable compared to the previous financial year at €625 million, representing 13.2% of Group revenue, as well as the operating income which amounts to €278 million, i.e. 5.9% of Group revenue.
Pascal Lemaire, Executive Vice President Finance at Servier: “The 2020/21 financial year was marked by an important milestone in our oncology strategy with the successful acquisition and integration of the Agios Pharmaceuticals’ oncology business. The revenue increase was driven by sales of our brand-name medicines in oncology (+34.9%) with a particularly strong performance for Tibsovo® in the United States and Onivyde® in Japan. Despite the negative impact of exchange rates and pressure on sales in China related to government reforms, the Group’s revenue increased by +4.3% at constant rates (+0.8% at current rates) compared to the previous year. The 2020/21 results demonstrate the strength of the Group and confirm its ability to successfully integrate new acquisitions. We are confident that we will achieve our 2025 goals and continue to serve patient needs across the world.”
Key figures (as of September 30, 2021)
Accelerated growth in oncology
The acquisition of Agios Pharmaceuticals’ oncology business demonstrates how the Group’s strategy in oncology is accelerating. Since the beginning of the 2020/21 financial year, Servier has allocated more than half of its R&D budget to the discovery and development of cancer treatments. The Group’s major investment in oncology is now reflected in the seven medicines now available to patients.
The Group’s brand-name medicine revenue in oncology amounted to €604 million compared to €448 million in 2019/20, representing a 34.9% increase. This strong increase is explained by the inclusion of Tibsovo®2 into the portfolio, resulting from the acquisition of Agios oncology (+€69 million), and sales of Onivyde® which doubled compared to the previous financial year. This performance is supported by Japan, where Nihon Servier initiated the marketing of Onivyde® in June 2020. Accordingly, the Japanese subsidiary recorded an 87.5% increase in revenue at current rates, reaching €90 million in 2020/21 compared to €48 million in 2019/20.
Sustained activity in cardiometabolism and significant growth in venous diseases
Sales of brand-name medicines in cardiometabolism amounted to €2.067 billion (i.e., 44% of Group revenue), down 3.2% at constant rates (-6.9% at current rates) compared to 2019/20 (€2.221 billion) due in particular to a slowdown in sales in China.
Daflon® is an important part of the Group’s growth. In 2020/21, sales increased by 13,6% at constant rates (+7.5% at current rates), up €473 million compared to €440 million in 2019/20, propelling Daflon® to the forefront of the Group’s brand-name medicines in terms of revenue. The Group has announced an investment of €100 million for its Oril Industrie site (Normandy, France) to double the production capacity of the active ingredient of Daflon® by 2023 in order to meet the global growing demand. With the new production unit, innovative manufacturing processes will be implemented as well as the introduction of a new synthesis route to improve the efficiency and environmental performance of the production of the active ingredient.
Group driven by international sales of brand-name medicines
The share of Group revenue generated outside the European Union remains stable compared to the previous financial year and represents more than half of the consolidated revenue, i.e., 51%. The Group has distributed over a billion boxes of medicines worldwide.
China remains the Group’s leading brand-name subsidiary, despite a 22.8% drop, which was due to government reforms, with a revenue of €353 million compared to €457 million in 2019/20.
The U.S. subsidiary is now the Group’s second-largest subsidiary, ahead of Russia (€247 million), with a revenue of €255 million, up 31.4% (€194 million in 2019/20). This performance is due, in particular, to the inclusion in the portfolio of Tibsovo® that is marketed in the United States and a result of the acquisition of the oncology business coming from Agios Pharmaceuticals.
An agile and cross-functional R&D model
Claude Bertrand, Executive Vice President Research & Development at Servier: “Servier is pursuing the transformation of its R&D activity to deliver a new molecular entity every 3 years, with an increased effort in oncology, as well as focused investments in neuroscience and autoimmune diseases, in line with the Group’s strategy. Our goal is to develop new medicines for patients with high unmet medical needs and for smaller populations. The recent acquisitions have significantly strengthened our pipeline as well as our innovation capabilities in oncology. We now have a biotechnology center of expertise in Denmark, enhanced by our investment in bioproduction in France, as well as a new R&D center in Boston in the United States which will open in the spring of 2022. The Servier Research and Development Institute in Paris-Saclay, which is expected to open in 2023, will be the heart of the Group’s global R&D organization.”
A strengthened and balanced pipeline
Pipeline at the end of December 2021
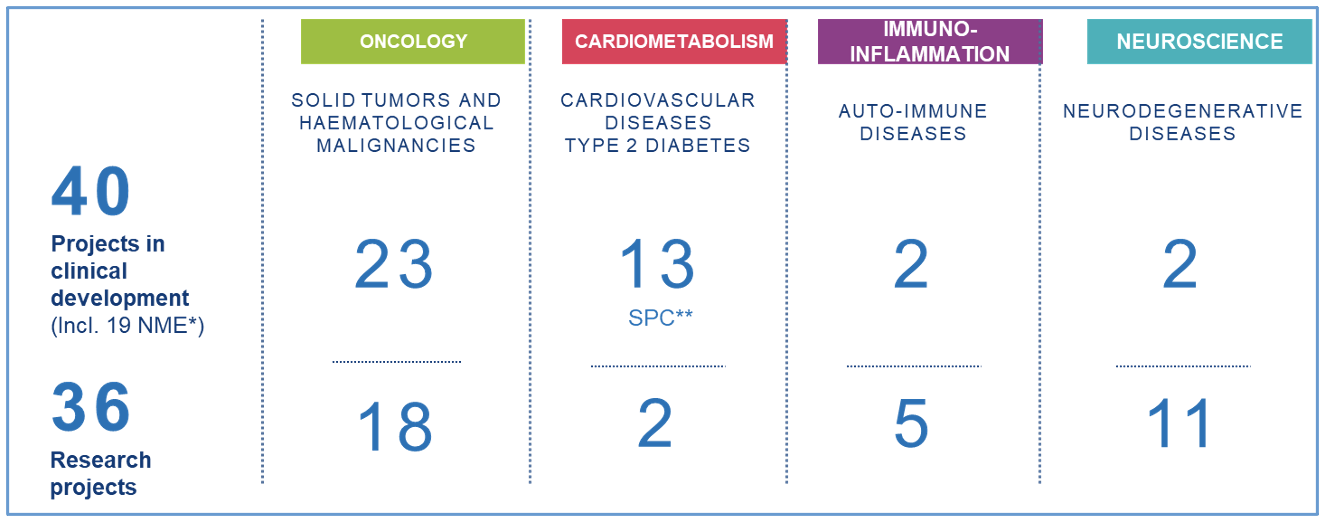
* New Molecular Entity
** Single Pill Combination – Fixed Combinations
With 40 projects in clinical development, including 19 new molecular entities, and 36 research projects1, resulting from significant and ongoing investment in R&D (more than 20% of brand-name revenue), Servier focuses its research and development efforts in therapeutic areas with high unmet medical need: oncology, neuroscience and immuno-inflammation. In cardiometabolism, Servier’s strategy is to pursue incremental innovation in order to continue to address patient needs. We focus on optimizing our existing medicines through, in particular, Single Pill Combinations (SPCs), medicines combined in a single tablet, which promote both a simpler treatment and better compliance.
Servier has placed oncology among its priorities and allocates more than 50% of its R&D budget to fighting cancer and addressing critical unmet patient needs. The Group focuses on cancers in specific populations that are difficult to treat, such as gastrointestinal, hematologic, pancreatic and pediatric cancers, and conducts its R&D programs using several approaches: apoptosis (or programmed cell death), immuno-oncology, with many research programs developed following the acquisition of Symphogen in 2020, and the metabolism of the cancer cell, an approach developed by teams from the oncology business of Agios Pharmaceuticals.
The acquisition of Agios Pharmaceuticals’ oncology business has significantly strengthened the oncology pipeline, with projects at all stages of research and development. Among the studies in phase 3, one on Tibsovo® (ivosidenib tablets), with the AGILE study4, was conducted in record time in two years, with positive results (see box) presented at the 63rd annual meeting of the American Society of Hematology (ASH). Additionally, vorasidenib, a dual inhibitor of IDH1 and IDH2 mutations that penetrates the brain, that is tested in the INDIGO phase 3 study in patients with IDH mutant low-grade glioma (brain tumor).
During this financial year, Servier has doubled the number of R&D programs in neuroscience and immuno-inflammation.
In neuroscience, Servier targets proteinopathies, characterized by the abnormal accumulation of specific proteins, as in Parkinson’s disease or amyotrophic lateral sclerosis.
The neuroscience pipeline includes two clinical development projects. The first is in partnership with Ose Immunotherapeutics on a potential treatment for Sjögren’s syndrome, an autoimmune disease characterized by lymphoid infiltration of the salivary and tear glands causing dry mouth and eyes. The second should start in 2022 and is in partnership with Oncodesign in Parkinson’s disease, a neurodegenerative disease characterized by the destruction of a specific group of neurons, dopamine neurons in the black substance of the brain. More than 6.3 million people worldwide are affected by Parkinson’s disease5.
An open and collaborative innovation dynamic
Servier is committed to open and collaborative innovation. The Group collaborates with a network of partners including pharmaceutical and biotech companies as well as academic institutions.
During the 2020/21 financial year, Servier entered new partnerships and reached significant milestones in collaborative studies with partners.
In oncology, Servier has signed two new partnerships, one with the American biotech company Celsius Therapeutics to pursue new therapeutic targets for the treatment of colorectal cancer (CRC), and the other with the Japanese biotechnology company PRISM BioLab in the field of immuno-oncology.
For instance, two important milestones have been achieved for the discovery of new therapeutic targets in oncology in research and in the preclinical phase as part of the partnership with the British laboratory Vernalis. Overall, Servier has 18 oncology partnerships6.
In neuroscience, Servier has established three new partnerships. The first is a research collaboration with Mina Therapeutics, a pioneering company in RNA activation therapies, in order to identify and develop therapies using small activating RNAs (saRNA). The second is a strategic collaboration with Nymirum, in order to identify and develop RNA-modulating drugs for the treatment of neurological diseases. Servier has also entered into a partnership with the company X-Chem to identify and develop new small molecules for the treatment of neurological disorders.
Additionally, Servier and Oncodesign, a French biotech specializing in precision medicine, announced in June :2021 the selection of a preclinical candidate from their collaboration in Parkinson’s disease. Working together since March 2019, the Group and the French biotech are leading the discovery of LRRK2 kinase inhibitors and their action as potential therapeutic agents against Parkinson’s disease.
In immuno-inflammation, a first patient was enrolled in August 2021 in the phase 2 clinical trial, conducted with the French biotechnology company Ose Immunotherapeutics, which evaluates the efficacy and safety of an antibody blocking the interleukin 7 receptor in Sjögren’s syndrome.
Finally, Servier has also partnered with Biolabs, an internationally recognized American incubator company, to manage the start-up incubator dedicated to new innovative companies in the health sector and located at the future Servier R&D Institute in Paris-Saclay.

The opening of the Research and Development Institute in Paris-Saclay is a major step in the transformation of Servier R&D demonstrating its ambition to facilitate a more open, more dynamic and more productive research for the benefit of patients. The Servier R&D Institute will work directly with the Group’s R&D centers, based in Denmark (Ballerup), the United States (Boston) and Hungary (Budapest), as well as the 15 international therapeutic research centers responsible for conducting clinical studies, and with its international network in charge of external innovation.
As of 2023, 1,500 R&D employees of the Group will be together at this new center, which will enable Servier to address the main challenges of the pharmaceutical industry today: discovering new therapeutic solutions for patients.
About Servier
Servier is a global pharmaceutical group governed by a Foundation. With a strong international presence in 150 countries and a total revenue of 4.7 billion euros in 2021, Servier employs 21,800 people worldwide. Servier is an independent group that invests over 20% of its brand-name revenue in Research and Development every year. To accelerate therapeutic innovation for the benefit of patients, the Group is committed to open and collaborative innovation with academic partners, pharmaceutical groups, and biotech companies. It also integrates the patient’s voice at the heart of its activities.
A leader in cardiology, the ambition of the Servier Group is to become a renowned and innovative player in oncology. Its growth is based on a sustained commitment to cardiovascular and metabolic diseases, oncology, neuroscience and immuno-inflammatory diseases. To promote access to healthcare for all, the Servier Group also offers a range of quality generic drugs covering most pathologies. More information: servier.com
- Source GERS, September 2021.
- Tibsovo® is currently approved in the United States as monotherapy for the treatment of adults with relapsed or refractory acute myeloid leukemia (AML) with IDH1 mutation and adults ≥ 75 years of age with newly diagnosed IDH1 mutation AML, or who have comorbidities preventing the use of intensive induction therapy.
- Data as of December 2021
- The AGILE trial is an international phase 3 clinical study, multicentric, double-blind, randomized, placebo-controlled, aimed at evaluating the efficacy and safety of the use of Tibsovo® in combination with azacitidine and comparing it to a placebo in combination with azacitidine, in adults with acute myeloid leukemia (AML) who are carriers of an IDH1 gene mutation and not eligible for intensive chemotherapy (≥ 75 years old or with comorbidities excluding intensive induction therapy).
- Source: European Brain Council: http://www.braincouncil.eu/library/disease-fact-sheets/parkinsons-disease/
- As of the end of December 2021